Brianne Dressen Severe Ongoing Adverse Reactions From AstraZeneca Clinical Trial of 2020

Brianne’s story:
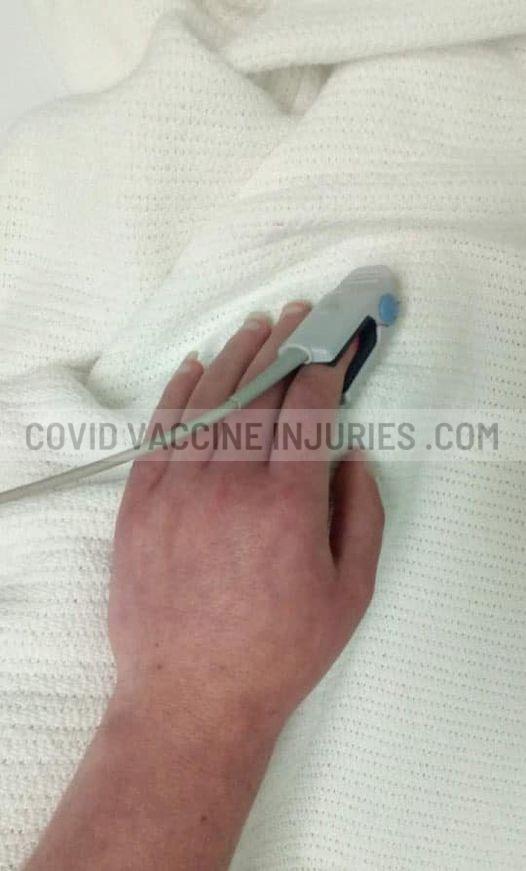
A previously healthy mother of two young children, who enrolled in the AstraZeneca clinical trial in the United States in November 2020.
“Immediately within an hour I had tingling down my arm…and by the time I got home, my vision was blurry and double. The following morning when I got up to go to work, the left side of my body was slumped over.”
AstraZeneca ran several tests, all of which came back negative for any neurodegenerative issues.
They sent her home, where her condition continued to decline. She has been hospitalized multiple times. At one point she was paralyzed from the waist down and had to relearn how to do a lot of basic body functions.
She was trapped inside her own body. Suffering.
Here is a news clip featuring her story.
Brianne’s husband, Brian Dressen, a Ph.D., provided the FDA his testimony below:
“Your decision is being rushed, based on incomplete data from underpowered trials, insufficient to predict rates of severe and long-lasting adverse reactions. I urge the committee to reject the EUA [Emergency Use Authorization] modification and direct Pfizer to perform trials that will decisively demonstrate that the benefits outweigh the risks for children. I understand firsthand the impact that you will or will not have with the decision you’re going to make today.”
“Because study protocol requires two doses, she was dropped from the trial, and her access to the study app deleted. Her reaction is not described in the recently released clinical trial report — 266 participants are described as having an adverse event leading to discontinuation, with 56 neurological reactions tallied.”
Brian Dressen also said:
“Injured support groups are growing. Memberships number into at least the tens of thousands. We must do better. Those injured in a trial are a critical piece of vaccine safety data. They are being tossed aside and forgotten. The FDA has known first-hand about her case and thousands of others. The FDA has also stated that their own systems are not identifying this issue and that VAERS is not designed to identify any multi-symptom signals. The system is broken.”
AstraZeneca was made aware of her injuries. She was dropped from the trial because her injuries were so severe from the first dose, that she couldn’t get two doses. AstraZeneca required 2 doses to participate, so they removed her, and all of her clinical trial information and injury information.
Brianne’s had to pay for her own medical debts associated with this injury.
Please have a listen to her Washington DC Testimony below, it’s extremely important to hear her experience. Please watch all the way to the end.
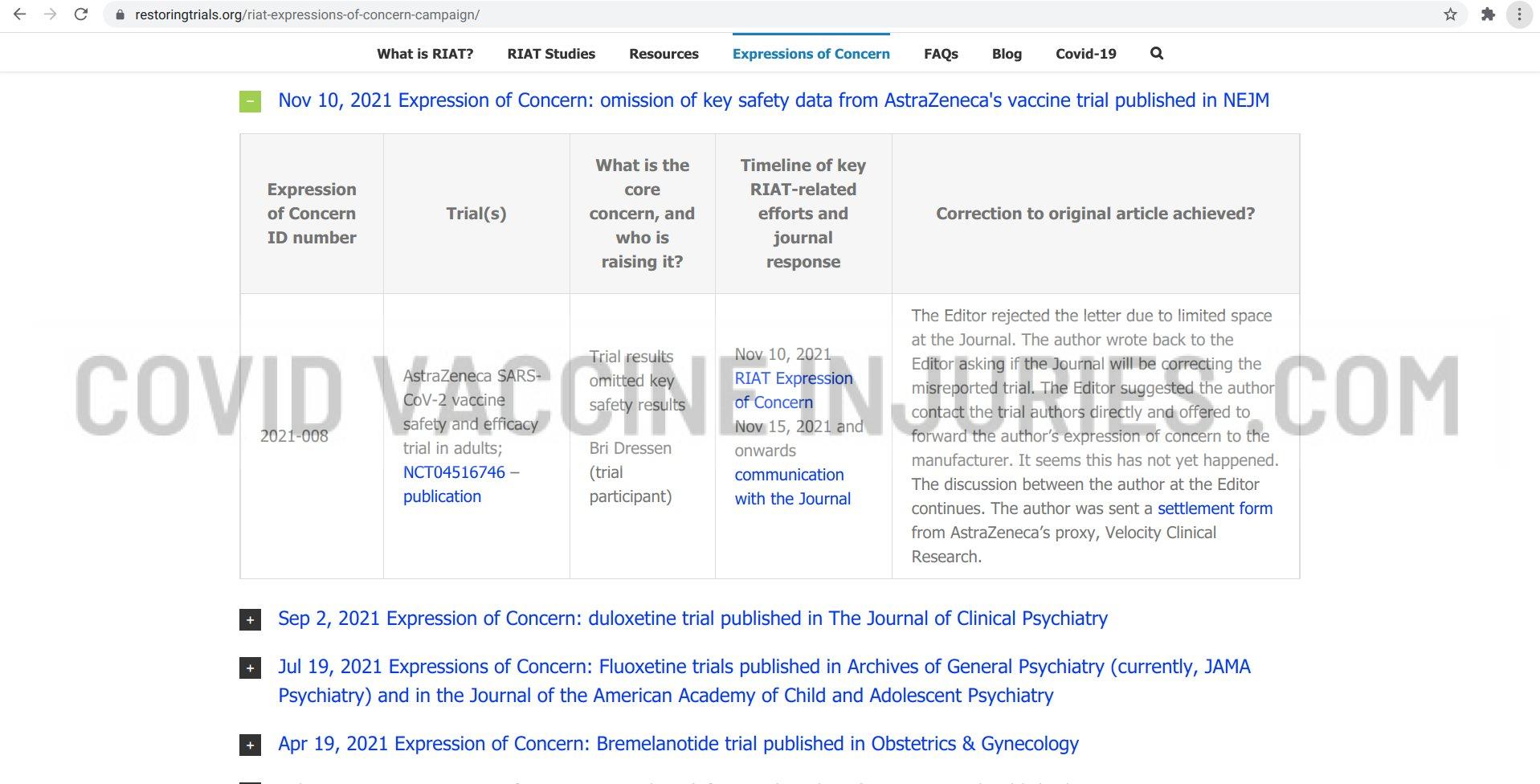
Following the Washington DC panel, Brianne wrote to the New England Journal of Medicine, as reported HERE.
Here are images of their response:
Here is a video from The Highwire on Brianne’s injuries:
You can watch the entire Washington DC Testimony including all of the other injury stories shared that day:















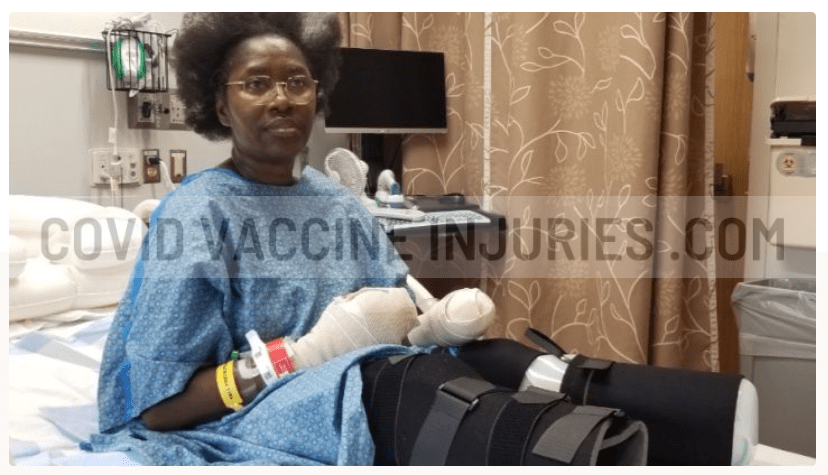
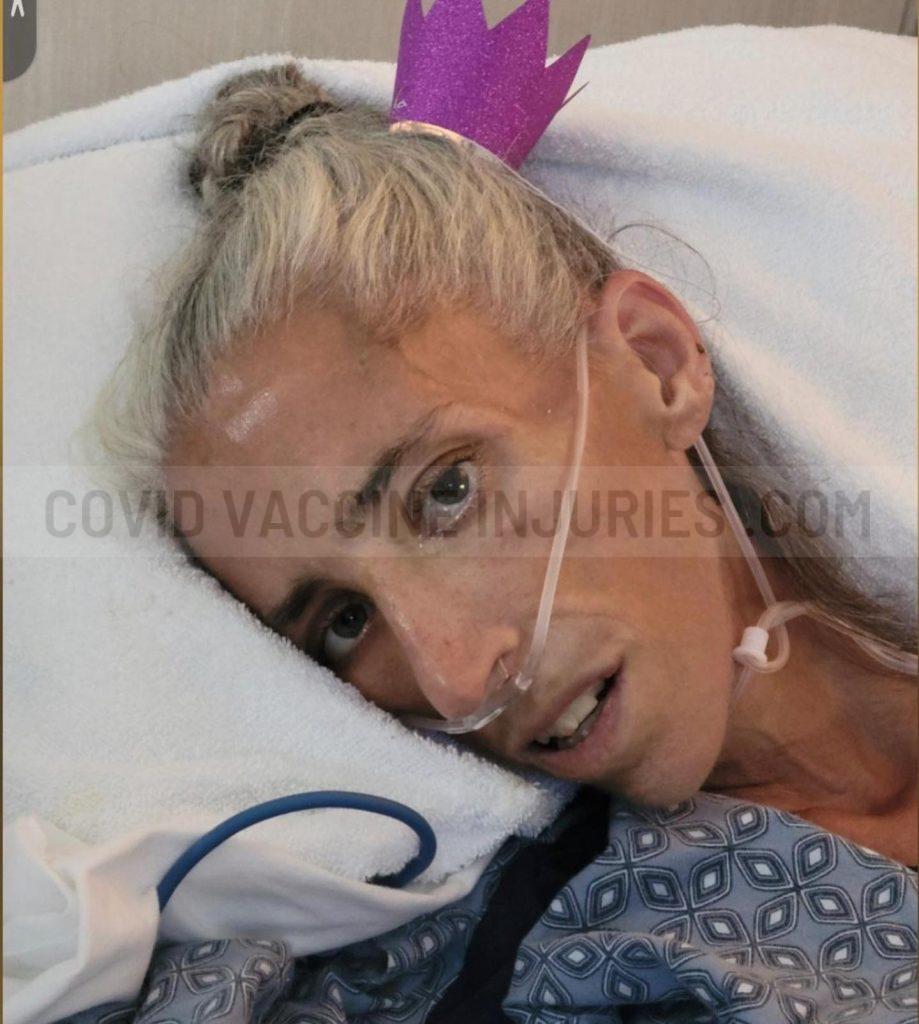





Responses